Researchers in CCIL Director Rohit Bhargava’s Chemical Imaging and Structures Laboratory (from left): Andres Orr, Rohit Bhargava, Matthew Confer, Rou-Jing Ho, Yen-Ting Liu, Ishaan Sharma, Kevin Yeh, and Kianoush Falahkheirkhah
Cancer Center at Illinois (CCIL) Director Rohit Bhargava’s Chemical Imaging and Structures Laboratory has developed a novel infrared microscopy system that dramatically improves the speed and quality of chemical imaging. The team’s microscope holds the potential to shift the landscape of current pathology, opening new frontiers in the study and diagnosis of diseases such as cancer. Unlike a common light microscope that only measures the structure of a sample, this microscope can also measure the chemistry within the sample.
“In my journey as an imaging scientist, this is a milestone development. This system offers the high performance we’ve been aiming for at a speed that is just not available anywhere else,” said Bhargava. “The imaging community has grappled for a long time with developing an instrument and imaging technology that is fast, reliable, robust, and acquires high quality, useful data. We solved that challenge in this study by going back to the basics. We started from our expertise in theory and modeling and re-thought the image formation process. This is a reimagining of the infrared microscope from the ground up and is culmination of years of effort by our large team to get that optimal instrument.”
Watch the CCIL’s interview with Rohit and his lab team, posted at the end of this story.
As reported in Nature Communications, the system is a mid-infrared confocal laser scanning microscope (IR-LSM) that enables label-free biomedical analysis of whole slide samples, providing high-throughput, low noise, and high spatial resolution images. When integrated with machine learning, the microscope facilitates efficient, comprehensive assessment of whole model organisms and histological assessments of disease in a manner compatible with the clinical environment. The benefits of mid-infrared spectroscopic imaging promise the highest optical molecular sensitivity due to light absorption at mid-IR frequencies resonating with molecular vibrational modes. Sophisticated analyses of these data, in tandem with recent progress in artificial intelligence (AI), greatly expands the potential scope and speed of comprehensive chemical imaging for scientific and biomedical applications.
Using chemical imaging in cancer pathology presented noteworthy challenges for Bharava’s research team over the years of developing their evolving instrument, which is now in its third iteration. For this milestone moment in the evolution of the imaging instrument, the team had two ambitious, simultaneous goals: speed and quality.
“The problem in acquiring infrared microscopy images is doing so quickly and with high fidelity,” said Kevin Yeh, who is a postdoctoral researcher with Bhargava, and first author of the publication. Yeh has led the development of multiple generations of infrared microscopes in the lab. Earlier generations were constructed from off-the-shelf and repurposed components and laid the groundwork for infrared microscope design with lasers. Building upon this research, members of the team designed and fabricated the microscope from custom optical components, hardware designed and tested at Illinois, and software they wrote. This careful engineering resulted in a microscope capable of collecting higher quality data with clinically viable throughput. “In this current iteration detailed in our paper, we merged what we’ve learned from our previous system designs to build a deployable platform for fast, high quality infrared microscopy that cancer clinicians and researchers will find easy to use,” explained Yeh.
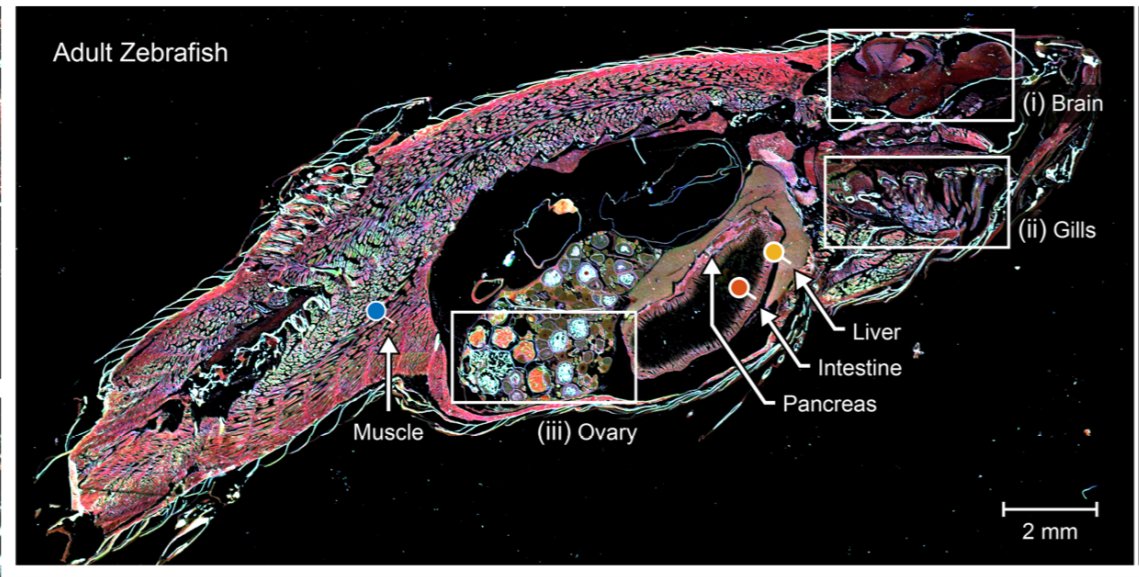
The team’s IR-LSM speed and data quality allows them to perform experiments and diagnostics they otherwise would not attempt, such as the chemical imaging of a whole model organism. The adult zebrafish sample above was imaged in approximately 15 minutes. Traditional imaging methods may have taken as long as 2-4 weeks.
The first order of business for Bhargava’s lab was to acquire data fast enough with both high chemical content and image quality. The second was to acquire the data with enough quantity that, when fed into an AI algorithm, the system gave desirable, dependable results. “For a very long time, cancer pathology has relied on looking at structural changes in tissues. This is important because sometimes chemical changes precede structural changes in tissue. And now, with the availability of artificial intelligence, we can use our large reservoir of chemical data together with AI to make better diagnoses,” said Bhargava.
The combination of chemistry and structure measured makes it ideal to use machine learning to extract information reliably and efficiently. With the assistance of pathology experts who can properly interpret the data, the team developed AI models to effectively extract cancer pathology information from the raw data. Postdoctoral researcher Kianoush Falahkheirkhah, a recent chemical engineering doctoral graduate, provided the leadership of the AI development. “We are changing the way we measure and acquire data or images of cancerous tissues and how we interpret that data. We want to use chemical imaging and look at the samples in a different way. We want to look at the chemical composition of the samples along with the spatial information. And that’s where AI can play an important role, because the data we acquire can be very helpful but not easily interpretable, unlike conventionally stained pathology samples. What we’re trying to do is to make that data interpretable. And that’s where the intersection of AI and our chemical imaging microscopes are coming from,” said Falahkheirkhah.

Post-doctoral researcher Kianoush Falahkheirkhah reviews the results of IR-LSM’s whole-slide image, produced in approximately two minutes.
The development of this instrument was possible due to the interdisciplinary strengths of a diverse team available at Illinois, bringing together mechanical and materials engineers; physics, biology, and chemical experts; software developers; and deep learning model systems researchers, as well as external clinical collaborators. “Another strength of this study is our clinical partnerships with Mayo Clinic and Carle Health,” said Bhargava. “At Mayo Clinic we worked with one of the leading pathologists in the world, Dr. John Chevelle, who has been a pioneer in diagnosing prostate cancer. Together, we found a workflow in which this technology could be useful in the clinic. And with Mayo Clinic resources, we were able to access a large number of samples, a large diversity of diseases, and a better understanding of how this technology could fit into the clinical workflow.”
The team’s second clinical collaboration is with Carle’s Dr. Georgina Cheng, who also provides expertise on the interoperative use of the team’s technology. “This dual clinical collaboration with Carle and Mayo Clinic assures us that our technologies are designed from the ground up to be successful in different health systems such as at Mayo and also in our local community,” said Bhargava.
What’s next for the Chemical Imaging and Structures Lab? “Now we need to apply this technology to many different diseases and then try to make more sophisticated decisions about the data,” explained Bhargava. “Can we find diseases other than cancer? Can we grade cancer? Can we use AI, together with the pathologist, to make more consistent, rapid, and more accurate decisions? These are the kinds of questions that this study enables, and which we are now exploring.”
“Our goal is for our AI models to be clinically relevant,” remarked Falahkeirkhah. “For us to be able to transition from research into the clinic, we need to increase the robustness and generalization of our models. We can’t limit ourselves to the set of samples we’ve been given. In the real world, on new samples with unknown diagnoses, it still needs to work. That’s the next step.”
Bhargava believes this is precisely the kind of study that exemplifies the unique powers available at Illinois, making the CCIL a unique and attractive place among the nation’s landscape of cancer research institutes. “This project represents the kind of study that our cancer center exists to undertake,” said Bhargava. “We bring in basic engineering, high-quality engineering at the University of Illinois and combine it with the strength of our clinical partners like Mayo and Carle. This combination helps us solve real problems. That’s what our cancer center is all about – taking basic engineering developments and bringing them rapidly to our clinical colleagues to make useful, high-impact decisions.”
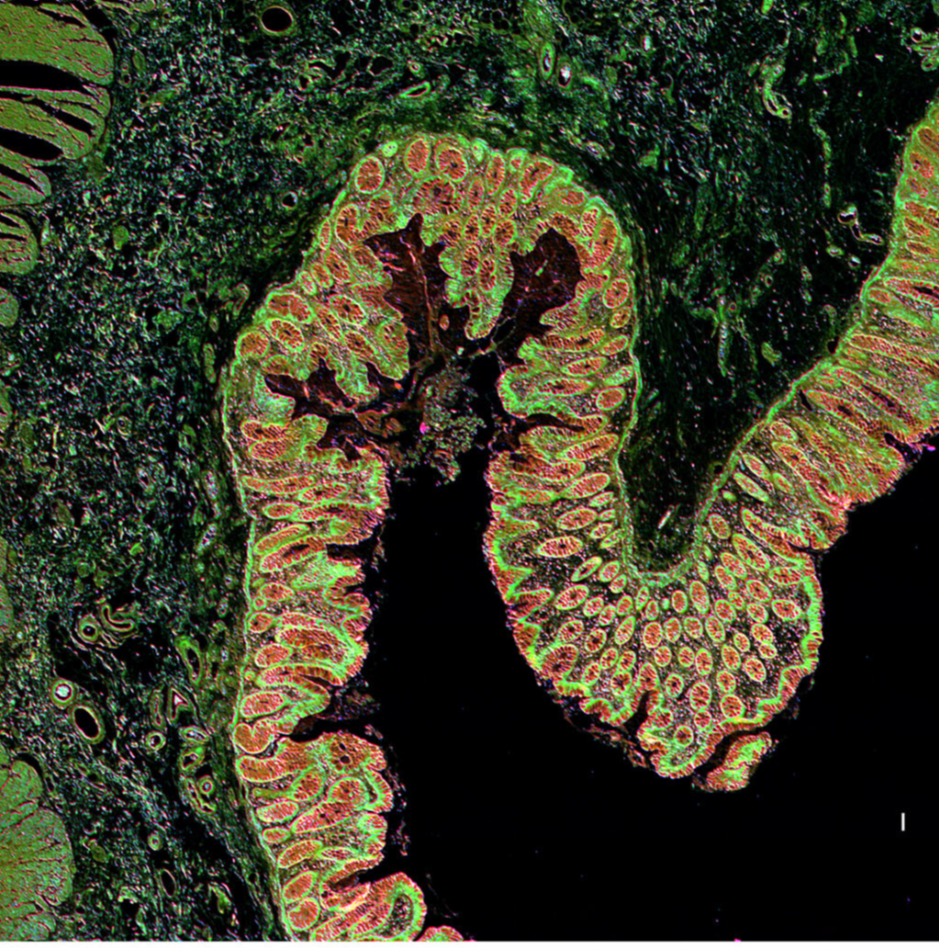
Visualization of colon tissue produced by the IR-LSM. The team’s label-free imaging uses the intrinsic contrast (in this case, chemical contrast) of a sample for visualization and analyses, without using dyes or stains to highlight components of interest.
Editor’s notes:
Rohit Bhargava is the Cancer Center at Illinois (CCIL) Director, Grainger Distinguished Chair in Bioengineering, and is affiliated with the Beckman Institute for Advanced Science and Technology.
This research was supported by funding from the National Institutes of Health, the Mayo Clinic, and the National Science Foundation.
To contact Rohit Bhargava, email rxb@illinois.edu
The paper “Infrared spectroscopic laser scanning confocal microscopy for whole-slide chemical imaging” is available online. https://doi.org/10.1038/s41467-023-40740-w
Written by Jonathan King, CCIL Communications Specialist

